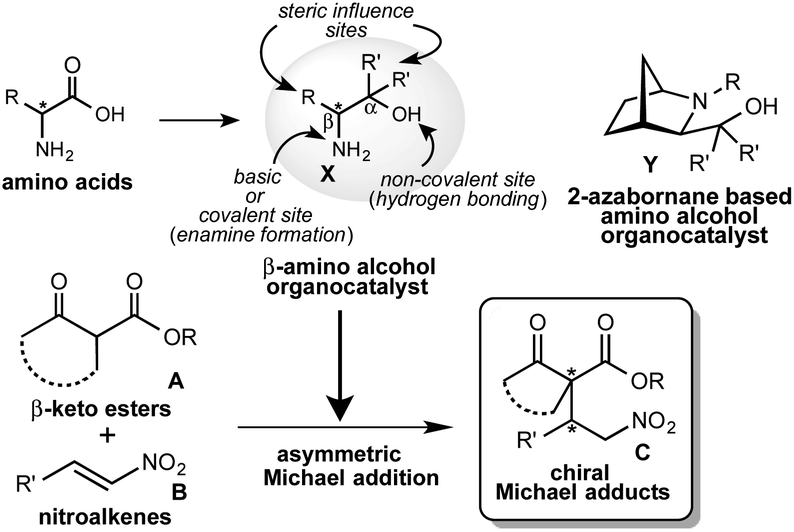
![PDF] A Brief Review on Synthesis of ò-amino Alcohols by Ring Opening ofEpoxides | Semantic Scholar PDF] A Brief Review on Synthesis of ò-amino Alcohols by Ring Opening ofEpoxides | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/874e9622a53f180f841e605dba48a23615d350be/2-Figure1-1.png)
PDF] A Brief Review on Synthesis of ò-amino Alcohols by Ring Opening ofEpoxides | Semantic Scholar

1,2-Amino Alcohols via Cr/Photoredox Dual-Catalyzed Addition of α-Amino Carbanion Equivalents to Carbonyls | Journal of the American Chemical Society
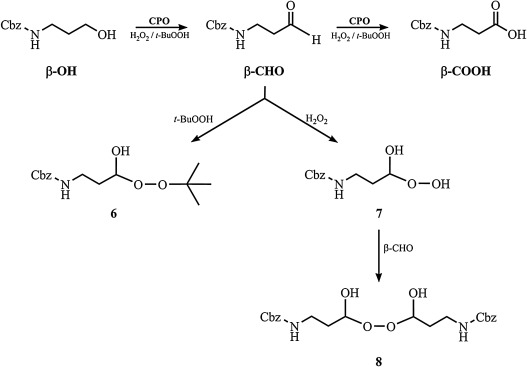
Chloroperoxidase-catalyzed amino alcohol oxidation: Substrate specificity and novel strategy for the synthesis of N-Cbz-3-aminopropanal | SeRMN – NMR Service at UAB
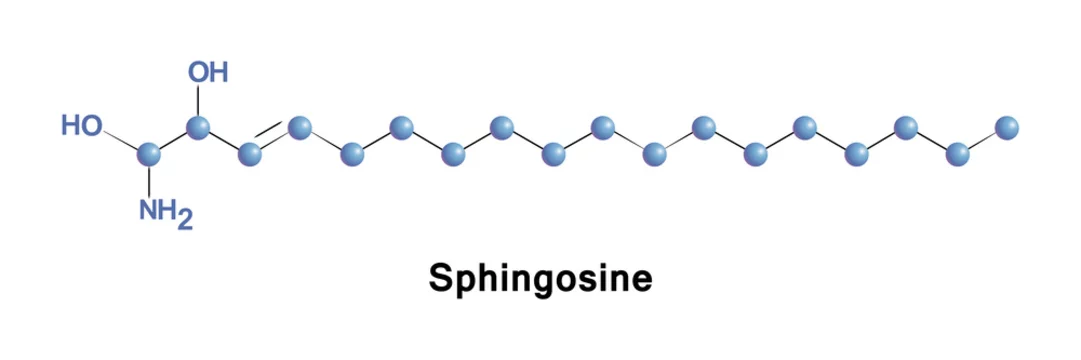
Sphingosine is an 18-carbon amino alcohol with an unsaturated hydrocarbon chain, forms a primary part of sphingolipids, a class of cell membrane lipids that include sphingomyelin, a phospholipid. Stock Vector | Adobe
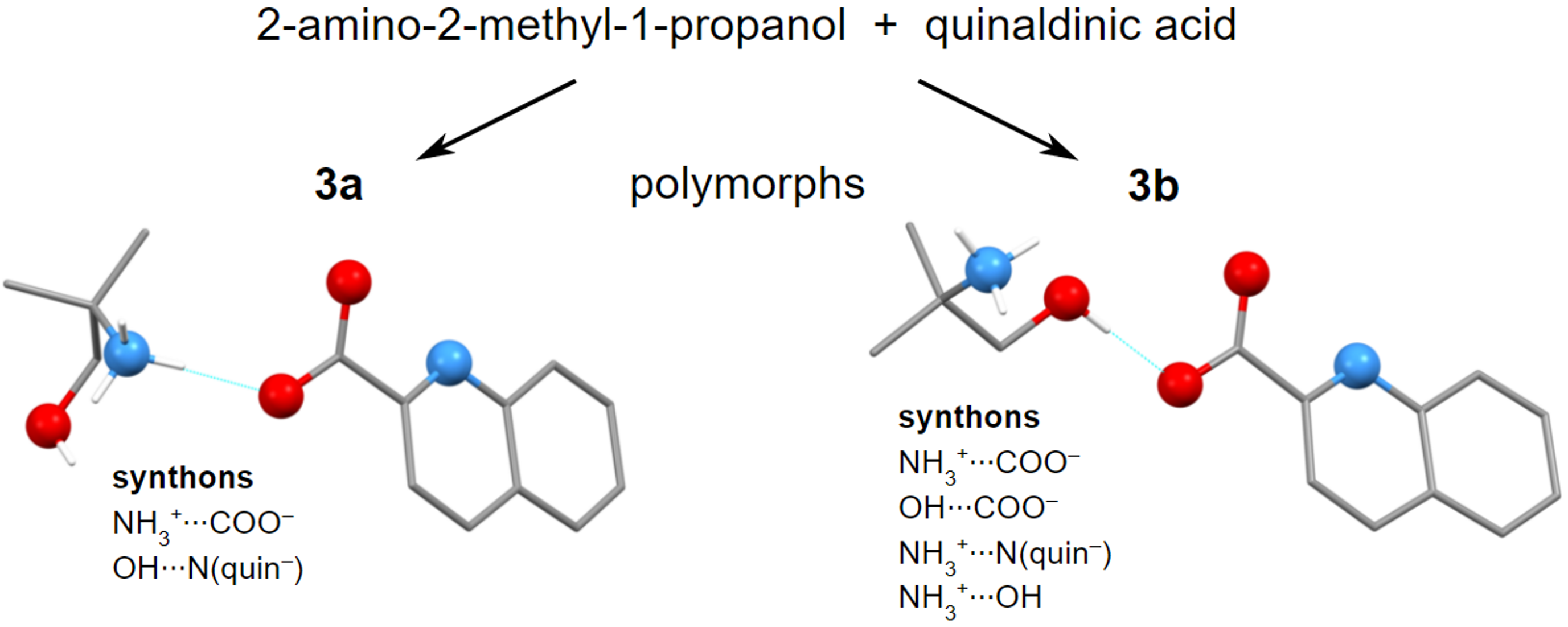
Molecules | Free Full-Text | Hydrogen Bonding and Polymorphism of Amino Alcohol Salts with Quinaldinate: Structural Study

Iridium-catalyzed enantioselective synthesis of chiral γ-amino alcohols and intermediates of (S)-duloxetine, (R)-fluoxetine, and (R)-atomoxetine | Communications Chemistry
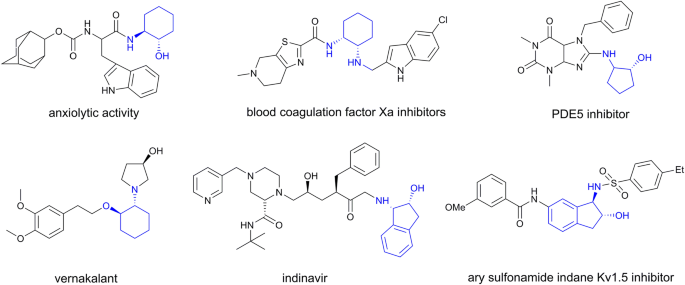
High throughput solid-phase screening of bacteria with cyclic amino alcohol deamination activity for enantioselective synthesis of chiral cyclic β-amino alcohols | SpringerLink

Amino alcohol monomers The amino alcohols that comprised oligomers A1... | Download Scientific Diagram

1,2-Amino Alcohols via Cr/Photoredox Dual-Catalyzed Addition of α-Amino Carbanion Equivalents to Carbonyls | Journal of the American Chemical Society

Synthesis of 1,3-Amino Alcohols, 1,3-Diols, Amines, and Carboxylic Acids from Terminal Alkynes | The Journal of Organic Chemistry